Quick Take:
- Policy: CBN Unified Portal + CDSCO Integrated IT Platform
- Authority: Central Bureau of Narcotics (Ministry of Finance) + CDSCO (Ministry of Health)
- Affects: Pharma manufacturers, importers, exporters, state drug regulators, patients
- Effective: CBN portal live since April 2023; CDSCO end-to-end platform in development
- Key Change: Manual licensing replaced with 24×7 faceless, contactless digital processes
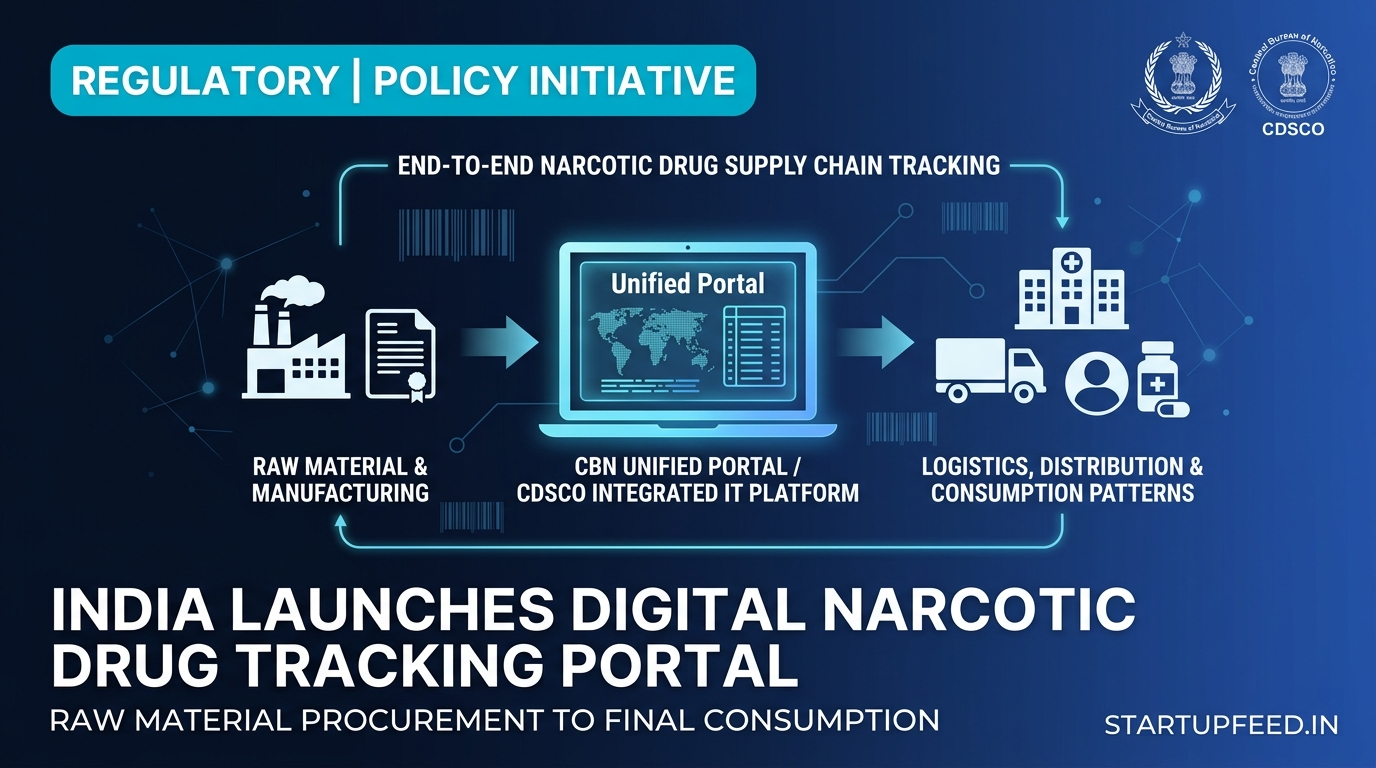
India has been laying the digital rails for full narcotics supply chain visibility — and the pieces are falling into place fast. The Unified Portal of the Central Bureau of Narcotics (CBN) was launched by Revenue Secretary Sanjay Malhotra as a one-stop solution for all licensing processes of the Bureau, including import certificates, export authorisations, manufacturing licences, and quota allocations for narcotic drugs and controlled substances. Alongside this, the apex drug regulator CDSCO has been working on an integrated IT platform that will track products right from the stage of procuring raw material, through the supply chain, and finally to patterns of consumption — closing the loop on end-to-end narcotics visibility for the first time.
This positions India’s drug control regime for a structural upgrade at a critical moment. With enforcement agencies struggling to trace diverted substances and counterfeit medicines proliferating, a connected digital backbone covering licensing, movement, and consumption is no longer optional — it is the floor.
StartupFeed Insight
What this means: India is stitching together a narcotics digital infrastructure layer that most developed nations still lack in integrated form — one that touches licensing, logistics, and consumption in a single traceable thread.
Winners:
- Compliant pharma manufacturers and exporters who will face faster licence turnaround and less paperwork
- Patients who stand to benefit from reduced drug diversion and counterfeit medicine exposure
- Law enforcement agencies gaining real-time data on drug movement
Losers:
- Entities currently exploiting manual process gaps for diversion — this architecture closes opacity
- Non-digitally ready smaller pharma players who may face compliance friction during the transition
Action required: Manufacturers, importers, and exporters of narcotic drugs and psychotropic substances should complete one-time registration on the CBN portal immediately. Watch for CDSCO integration notifications for the broader supply chain tracking mandate.
What’s New
The CBN Unified Portal is designed as an interface between the department and industry stakeholders, with the purpose of disseminating relevant information and digitising the Import/Export licence issuance process. The IT-enabled system introduces rules and policy to achieve transparent public delivery services, transforming physical processes into an online system with integration across other government applications.
All operations are faceless and contactless. Applicants can file applications from anywhere, at any time, on a 24×7 basis, eliminating the need for physical interaction. Queries with the department are raised and responded to through the portal itself.
The broader ambition extends well beyond licensing. The integrated IT platform CDSCO has been developing will be able to track products right from raw material procurement to supply chain movement to final consumption patterns. This effectively means the government is building a full-chain digital ledger for narcotics — from cultivation quota to patient.
Key Provisions
| Provision | Old System | New System | Impact |
| Import/Export Licensing | Manual, paper-based | Online, 24×7 digital portal | Faster turnaround, lower compliance cost |
| Manufacturing/Quota Licence | Physical applications | Digital submission + DSC | Faceless, contactless approvals |
| Supply Chain Tracking | No integrated system | End-to-end IT platform (CDSCO) | Raw material to consumption visibility |
| Inter-agency Data Sharing | Siloed systems | API integration with GST, PAN, UIDAI, e-Sanchit | Real-time cross-verification |
| Document Submission | Repeated per application | One-time registration with stored profile | Reduces duplication, saves time |
Who’s Affected
The scope covers every player in India’s legal narcotic drugs chain. Exporters, importers, and manufacturers of narcotic drugs, psychotropic substances, and controlled substances are the primary beneficiaries — with the portal simplifying obtaining import certificates, export authorisations, NOCs, manufacturing licences, and narcotic drug quota allocations in smooth, hassle-free operations.
State drug regulators and enforcement agencies benefit equally. Several digital initiatives including the NCORD portal, the Crime and Criminal Tracking Network System (CCTNS), and the National Integrated Database on Arrested Narco-Offenders (NIDAAN) are already in operation to strengthen drug law enforcement, intelligence sharing, investigation, and capacity building. The new portal layers commercial supply chain tracking onto this enforcement backbone.
The gap being plugged is real and urgent. Approximately 30% of seized drugs in India are not tracked, with no data on their source or distribution networks — reflecting serious data management and coordination loopholes.
Impact by Sector
| Sector | Impact | Why |
| Pharma Manufacturers | Positive | Faster licences, digital quota allocations, reduced paperwork |
| Exporters / Importers | Positive | 24×7 applications, API-linked verification cuts delays |
| State Drug Regulators | Positive | Real-time data sharing eliminates information silos |
| Law Enforcement (NCB, ANTFs) | Positive | Supply chain data strengthens diversion investigation |
| Small / Informal Distributors | Cautionary | Manual workarounds close; non-compliance risk rises |
| Patients | Positive | Reduced counterfeit drug risk, better essential medicine availability |
Compliance Checklist — Action Items for Regulated Entities
- Register on the CBN Unified Portal at cbnonline.gov.in — immediately
- Complete one-time profile with company PAN, GST, UIDAI-linked details
- Migrate all pending Import/Export licence applications to the digital system
- Monitor CDSCO Sugam portal for integration notifications on the supply chain tracking mandate
- Ensure internal ERP systems are API-compatible for eventual data sharing requirements
Industry Reaction
The launch was attended by representative stakeholders from pharma industries. The portal is calibrated to have database integration with Bharat Kosh, GST, PAN-NSDL validation, e-Sanchit, and UIDAI — facilitating single-point services for obtaining licences from CBN.
State governments are moving in parallel. Maharashtra’s Food and Drug Administration has proposed a statewide ‘Track and Trace’ system to the Central government to digitally monitor medicines across the supply chain, seeking Rs 26.5 Cr for the initiative. Haryana has directed all government hospitals to maintain medicine stock records on a real-time centralised portal, ensuring transparency and eliminating the need for patients to purchase medicines from outside.
What’s Next
India’s narcotic drug digital infrastructure is converging from three directions simultaneously — the CBN portal handling licensing, the CDSCO platform targeting end-to-end supply chain visibility, and state-level Track and Trace initiatives filling regional gaps. The question is not whether full-chain digital tracking arrives, but how fast the CDSCO integrated platform rolls out to cover the entire supply chain in a live, verifiable system.
The next 18 months will likely see CDSCO accelerate its integrated platform after years of development — particularly as counterfeit drug cases and diversion incidents put political pressure on regulators. Pharma companies that invest in compliance infrastructure now will gain a first-mover advantage when mandatory digital reporting expands.
Will India build the world’s most comprehensive narcotics supply chain tracking system — or will inter-ministry coordination gaps slow the rollout? That answer will define the effectiveness of every drug enforcement effort that follows. — Follow @StartupFeed_in for updates.